Density and Buoyancy are two aspects of science which affect fluids. Both Density and Buoyancy will affect the studies which are performed on a fluid. Density is the comparison between the mass and volume of an object. Basically, density depends on the mass of the atoms which make up the material of the object. Buoyancy on the other hand, is how something or someone will float. But, these two are intertwined, as you can see in the following.
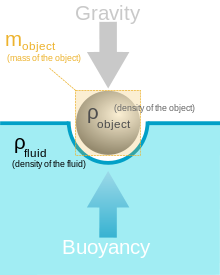
As you can see in the image above, the reason why the object is floating is because the upward force of Buoyancy is equal to the downward force of gravity, which is 9.8 m/s^2, therefore the force of Buoyancy must be 9.8 m/s^2. The reason why the two forces are equal is because the water that the object is floating in has a density of 1 g/cm^3, meaning that there is no resistance on the force of buoyancy. But, if you were to use multiple fluids with different densities... I wonder what would happen?
As you can see in the video above, when he puts the ball in, he first adds water to the cup, meaning that the ball will float, and have buoyancy. But, in his second beaker, he has a fluid called ethanol which has a lower density than water, causing a change of medium meaning that the ball doesn't have buoyancy anymore.
As you can see in this image, the boy is floating and has inflatable cushions around his arms. The reason why the child is able to float is because the "floaties" that he has on are buoyant, meaning that because the boy has low mass, the armbands are able to keep him up and floating. Many people use this, but few know how it works.
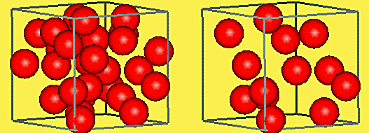
In this photo representing density, you can find that the image on the left is a more dense object. This is because the atoms inside the object are more plentiful, and very close together. This causes a high density, and usually objects with high density have problems being buoyant.
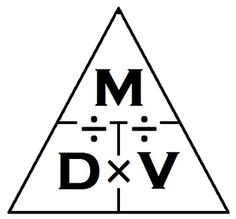
This image here represents the formula which we can use to find out Density. In this pyramid here, we have 3 formulas. The first one being to calculate Density, you would perform M (mass) / V (volume). The second formula which we have is what we can use when we require the volume of something, but we have the mass and Density. The formula which we would put into action would be M (mass) / D (density). Finally, our last formula will be used to find mass when we have the volume and the density. What we would do is just multiply D and V. The way that Density is measure is in g/cm^3, because mass is measured in grams, and then you divide by cm^3.
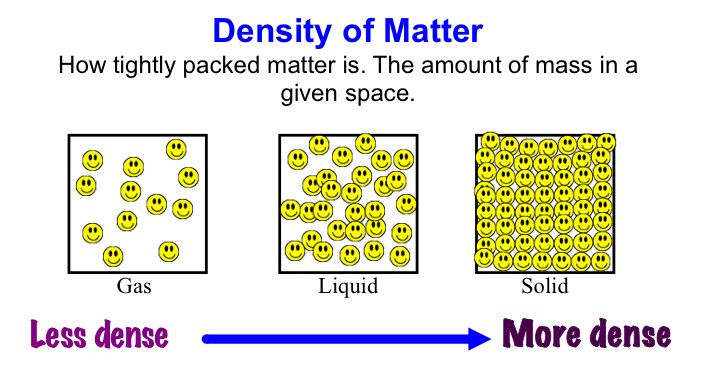
In this photo, we can see that Gases have substantially less atoms and mass than both liquids and solids. But, we can see that solids have a very tightly packed density, meaning that all the atoms are very close together. The reason why gases are not very dense is because they are not containable using regular methods. Also, gases move freely, and are not confined to a certain space. Liquids are roughly the same thing. Liquids will adapt to anything that they are put in, but are still not contained. Therefore, solids must have the most tightly packed density. This is because Density is confined, and thus the particles must be tightly packed or else you could just go straight through any empty spaces with no atoms.